Home
Products
Nagilactone B
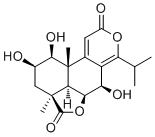


| Product Name | Nagilactone B |
| Price: | Inquiry |
| Catalog No.: | CN08504 |
| CAS No.: | 19891-51-1 |
| Molecular Formula: | C19H24O7 |
| Molecular Weight: | 364.4 g/mol |
| Purity: | >=98% |
| Type of Compound: | Diterpenoids |
| Physical Desc.: | Powder |
| Source: | The herbs of Podocarpus neriifolius |
| Solvent: | Chloroform, Dichloromethane, Ethyl Acetate, DMSO, Acetone, etc. |
| SMILES: |
| Contact us | |
|---|---|
| First Name: | |
| Last Name: | |
| E-mail: | |
| Question: | |
| Description | Nagilactone B, extracted from the root bark of Podocarpus nagi, is a liver X receptor (LXR) agonist. |
| Target | LXR[1] |
| In Vitro | RAW264.7 cells are co-incubated with oxLDL (20 μg/mL) and Nagilactone B (0.02, 0.1, and 0.5 μM) for 24 h. Oil Red O (ORO) staining reveals significant lipid accumulation and foam cell formation in RAW264.7 cells following oxLDL treatment. Nagilactone B (NLB) significantly ameliorates intracellular lipid accumulation. ORO positive areas are reduced by 30.05±7.49 (P<0.01), 47.25±5.39 (P<0.001), and 48.65±7.44% (P<0.001) in Nagilactone B (0.02, 0.1, and 0.5 μM)-treated groups, respectively. The effects of Nagilactone B are evaluated ton cholesterol efflux. Nagilactone B (0.02, 0.1, and 0.5 μM) markedly promotes cholesterol efflux to extracellular apolipoprotein A-I (apoA-I) and high density lipoprotein (HDL) with maximal 5.72- (P<0.05) and 2.34-fold (P<0.01), respectively[1]. |
| In Vivo | Nagilactone B (NLB) suppresses atherosclerosis in apoE-/- mice by inducing ATP-binding cassette transporter A1 (ABCA1) and G1 (ABCG1) mediated cholesterol efflux in macrophages. Male apoE-deficient mice on C57BL/6J background receive Nagilactone B (10 and 30 mg/kg) for 12 weeks. Compared with the model group, Nagilactone B treatment (10 and 30 mg/kg) significantly reduces en face lesions of total aorta areas. Six-week-old male apoE-/- mice on an HFD are randomized to receive Atorvastatin (10 mg/kg/day), Nagilactone B (10 and 30 mg/kg/day), or CMC-Na for 12 weeks. Mice on chow diet are administered CMC-Na as the normal diet control group. En face aortic lesion areas are evaluated with Sudan IV staining and lesion areas in the aortic sinus monitored via ORO staining. Atherosclerosis developes slowly in the normal diet group, whereas lesions in the HFD model group are significantly increased in en face aortas. Nagilactone B treatment (10 and 30 mg/kg/day) significantly reduces en face aortic lesions, compared with the HFD group by 54.96±10.06% (P<0.01), 71.50±15.37% (P<0.001) in both NLB (L) and NLB (H) groups. In particular, Nagilactone B markedly attenuates atherosclerotic plaque lesion areas in the aortic arch aorta, thoracic aorta, and abdominal aorta [P<0.01 in NLB (H) group][1]. |
| Cell Assay | RAW264.7 cells are plated into 12-well plates at 3×105 cells per well. After 12 h, cells are co-incubated with oxLDL (20 μg/mL) with or without Nagilactone B (0.02, 0.1, 0.5 μM) in DMEM for 24 h. At the end of the treatment period, cells are washed twice with PBS, fixed with 4% paraformaldehyde, and stained by filtered 0.5% ORO. Images are obtained via DP70 microscopy from at least six randomly selected fields for each condition. Data are analysed with Image-Pro Plus 6.0 software. The ratio of foam cells is calculated as the percentage of stained to total cell area[1]. |
| Animal Admin | Mice[1] ApoE-/- mice at 6 weeks of age on a C57BL/6J background are randomly divided into five groups [normal diet, model, Atorvastatin, NLB (L), and NLB (H) groups]. Atorvastatin and NLB are dissolved in CMC-Na before administered. Mice on the normal diet receive carboxymethylcellulose sodium (CMC-Na) as the negative control group. Mice on a high-fat diet (HFD) (0.15% cholesterol and 18% fat) are administered CMC-Na or Atorvastatin (10 mg/kg) or Nagilactone B [10 and 30 mg/kg, as NLB (L) and NLB (H), respectively] daily in an intragastric manner for 12 weeks. Body weight and food intake are monitored[1]. |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 644.6±55.0 °C at 760 mmHg |
| Flash Point | 233.7±25.0 °C |
| Exact Mass | 364.152191 |
| PSA | 117.20000 |
| LogP | -0.18 |
| Vapour Pressure | 0.0±4.3 mmHg at 25°C |
| Storage condition | 2-8℃ |