Home
Products
Metformin HCl
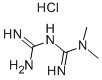


| Product Name | Metformin HCl |
| Price: | $23 / 20mg |
| Catalog No.: | CN00306 |
| CAS No.: | 1115-70-4 |
| Molecular Formula: | C4H11N5.HCl |
| Molecular Weight: | 165.62 g/mol |
| Purity: | >=98% |
| Type of Compound: | Alkaloids |
| Physical Desc.: | Powder |
| Source: | |
| Solvent: | Chloroform, Dichloromethane, Ethyl Acetate, DMSO, Acetone, etc. |
| SMILES: | CN(C(=N)NC(=N)N)C.Cl |
| Contact us | |
|---|---|
| First Name: | |
| Last Name: | |
| E-mail: | |
| Question: | |
| Description | Metformin (hydrochloride) is an FDA approved first-line drug for the treatment of type 2 diabetes. Metformin decreases hepatic glucose production, mostly through a mild and transient inhibition of the mitochondrial respiratory-chain complex 1. |
| Target | AMPK Autophagy Mitophagy |
| In Vitro | Metformin inhibits proliferation of ESCs in a concentration-dependent manner. The IC50 is 2.45 mM for A-ESCs and 7.87 mM for N-ESCs. Metformin shows pronounced effects on activation of AMPK signaling in A-ESCs from secretory phase than in cells from proliferative phase[3]. Metformin (0-500 μM) decreases glycogen synthesis in a dose-dependent manner with an IC50 value of 196.5 μM in cultured rat hepatocytes[4]. Metformin shows cell viability and cytotoxic effects on PC-3 cells with IC50 of 5 mM[5]. |
| In Vivo | Metformin (100 mg/kg, p.o.) alone, and metformin (25, 50, 100 mg/kg) with isoproterenol groups attenuates myocyte necrosis through histopathological analysis[1]. Metformin (> 900 mg/kg/day, p.o.) results in moribundity/mortality and clinical signs of toxicity in Crl:CD(SD) rats[2]. |
| Cell Assay | ESCs are plated in 96-well plates at a concentration of 1×103cells/well. After attachment, cells are treated with different doses of metformin/compound C for 0 min, 15 min, 1 h, and 24 h. MTT assays are performed as described previously. In brief, MTT (5 mg/mL) is added to the 96-well plates at a volume of 10 μL/well, and the plates are incubated for 4 h. The MTT reaction is terminated by removal of the culture medium containing MTT, and 100 μL DMSO per well are added and incubated at RT on a shaker for 10 min to ensure that the crystals had dissolved sufficiently. Absorbance values are measured at 595 nm. Cell proliferation (percentage of control) is calculated as follows: absorbance (experimental group)/absorbance (control group). Cell proliferation inhibition (percentage of control) is calculated as follows: 100%−cell proliferation (percentage of control). Each experiment is performed in duplicate and repeated six times to assess result consistency. |
| Animal Admin | The animals are randomized into six groups consisting of six rats each. Rats in group 1 (control) receives a subcutaneous injection of physiological saline (0.5 mL) and are left untreated for the entire experimental period. Rats in group 2 receives an oral administration of metformin (100 mg/kg; twice daily) for 2 days and are subcutaneously injected with saline at an interval of 24 h for 2 consecutive days. Rats in group 3 (MI control) receives an oral administration of saline (twice daily) for 2 days and are sc injected with isoproterenol (100 mg/kg) daily for 2 consecutive days at an interval of 24 h. Rats in groups 4 to 6 are treated with metformin at 25, 50, and 100 mg/kg. Metformin is dissolved in saline and is gavaged at a volume of 0.25-0.5 mL twice a day at an interval of 12 h, started immediately before isoproterenol injection. |
| Boiling Point | 224.1ºC at 760 mmHg |
| Flash Point | 89.3ºC |
| Exact Mass | 165.078125 |
| PSA | 88.99000 |
| LogP | 1.05850 |
| Vapour Pressure | 0.0929mmHg at 25°C |
| Storage condition | Store at RT |